General Information
-
DRAMP ID
- DRAMP02272
-
Peptide Name
- PGLa (chain of PYLa/PGLa A; Frogs, amphibians, animals)
-
Source
- Xenopus laevis (African clawed frog)
-
Family
- Belongs to the gastrin/cholecystokinin family (Magainin subfamily)
-
Gene
- pgla-a AND pgla-b
-
Sequence
- GMASKAGAIAGKIAKVALKAL
-
Sequence Length
- 21
-
Protein Existence
- Protein level
Activity Information
-
Biological Activity
- Antimicrobial, Antibacterial, Anti-Gram+, Anti-Gram-, Antifungal
-
Target Organism
-
- Gram-negative bacteria: Escherichia coli 25922 (MIC=62-125 µg/ml), Klebsiella pneumoniae 13883 (MIC=250-500 µg/ml), Pseudomonas aeruginosa 27853 (MIC=250-500 µg/ml);
- Gram-positive bacteria: Staphylococcus aureus 29213 (MIC=125-500 µg/ml), Strepococcus faecalis (MIC>500 µg/ml).
- Yeast: Candida albicans 14053 (MIC=250-500 µg/ml).
-
Hemolytic Activity
-
- No hemolysis information or data found in the reference(s) presented in this entry
-
Cytotoxicity
-
- Not included yet
-
Binding Target
- Not found
Structure Information
-
Linear/Cyclic
- Not included yet
-
N-terminal Modification
- Not included yet
-
C-terminal Modification
- Not included yet
-
Nonterminal Modifications and Unusual Amino Acids
- Not included yet
-
Stereochemistry
- Not included yet
-
Structure
- Not found
-
Structure Description
- Not found
-
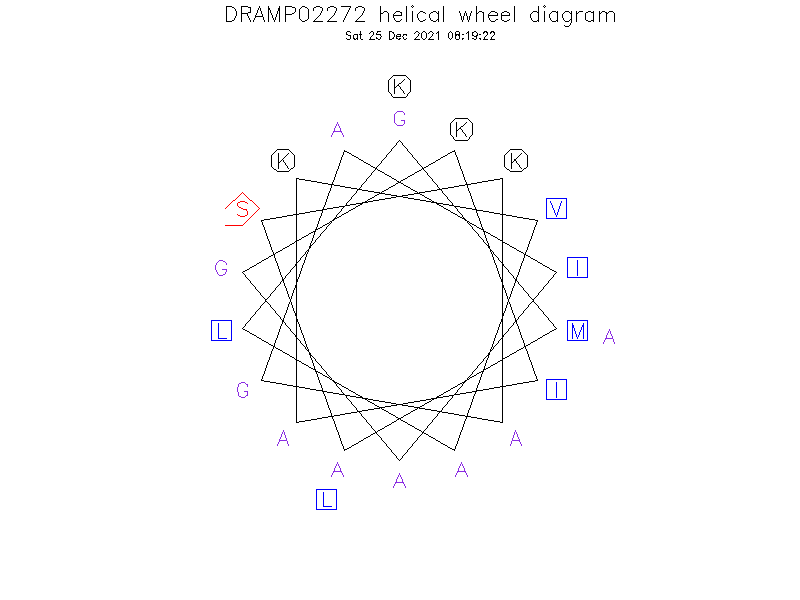
Helical Wheel Diagram
-
PDB ID
- None
-
Predicted Structure
- There is no predicted structure for DRAMP02272.
Physicochemical Information
-
Formula
- C88H161N25O23S
Absent Amino Acids
- CDEFHNPQRTWY
Common Amino Acids
- A
Mass
- 1969.46
PI
- 10.48
Basic Residues
- 4
Acidic Residues
- 0
Hydrophobic Residues
- 12
Net Charge
- +4
-
Boman Index
- 15.96
Hydrophobicity
- 0.843
Aliphatic Index
- 121.43
Half Life
-
- Mammalian:30 hour
- Yeast:>20 hour
- E.coli:>10 hour
Extinction Coefficient Cystines
- 0
Absorbance 280nm
- 0
Polar Residues
- 4
DRAMP02272
Comments Information
Function
- PGLa display a broad-spectrum of antibacterial activity against a range of Gram-positive and Gram-negative bacteria. PGLa also displays antifungal activity against C. albicans ATCC 14053.
Tissue specificity
- Expressed by the skin glands. Synthesized in the stomach and stored in a novel granular multinucleated cell in the gastric mucosa. Stored as active, processed peptides in large granules within the granular gland secretions of the skin.
PTM
- Leucine amide at position 21.
Literature Information
- ·Literature 1
-
Title
- Biosynthesis and degradation of peptides derived from Xenopus laevis prohormones.
-
Pubmed ID
- 3606567
-
Reference
- Biochem J. 1987 Apr 1;243(1):113-120.
-
Author
- Giovannini MG, Poulter L, Gibson BW, Williams DH.
- ·Literature 2
-
Title
- Antimicrobial peptides in the stomach of Xenopus laevis.
-
Pubmed ID
- 1717472
-
Reference
- J Biol Chem. 1991 Oct 15;266(29):19851-19857.
-
Author
- Moore KS, Bevins CL, Brasseur MM, Tomassini N, Turner K, Eck H, Zasloff M.