General Information
-
DRAMP ID
- DRAMP18443
-
Peptide Name
- Smp24 (scorpions, arachnids, Chelicerata, arthropods, invertebrates, animals)
-
Source
- venom, Scorpio maurus palmatus
-
Family
- Not found
-
Gene
- Not found
-
Sequence
- IWSFLIKAATKLLPSLFGGGKKDS
-
Sequence Length
- 24
-
UniProt Entry
- No entry found
-
Protein Existence
Activity Information
-
Biological Activity
- Antimicrobial, Antibacterial, Antifungal
-
Target Organism
- Active against B. subtilis NCIMB 8024, S. epidermidis sp., S. aureus SH100 (MIC 4-8 ug/ml), E. coli JM109 (MIC 64 ug/ml), and C. albicans (MIC 32 ug/ml).
-
Hemolytic Activity
-
- No hemolysis information or data found in the reference(s) presented in this entry
-
Cytotoxicity
-
- Not included yet
-
Binding Target
- Cell membrance
Structure Information
-
Linear/Cyclic
- Not included yet
-
N-terminal Modification
- Not included yet
-
C-terminal Modification
- Not included yet
-
Nonterminal Modifications and Unusual Amino Acids
- Not included yet
-
Stereochemistry
- Not included yet
-
Structure
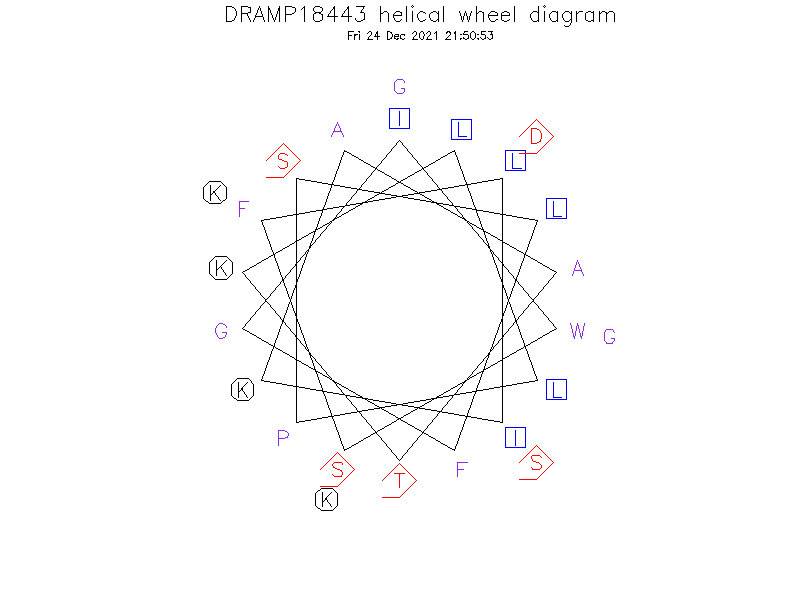
- Helix
-
Structure Description
- Not found
-
Helical Wheel Diagram
-
PDB ID
- None
-
Predicted Structure
- There is no predicted structure for DRAMP18443.
Physicochemical Information
-
Formula
- C123H197N29O31
Absent Amino Acids
- CEHMNQRVY
Common Amino Acids
- KL
Mass
- 2578.09
PI
- 10
Basic Residues
- 4
Acidic Residues
- 1
Hydrophobic Residues
- 11
Net Charge
- +3
-
Boman Index
- 56
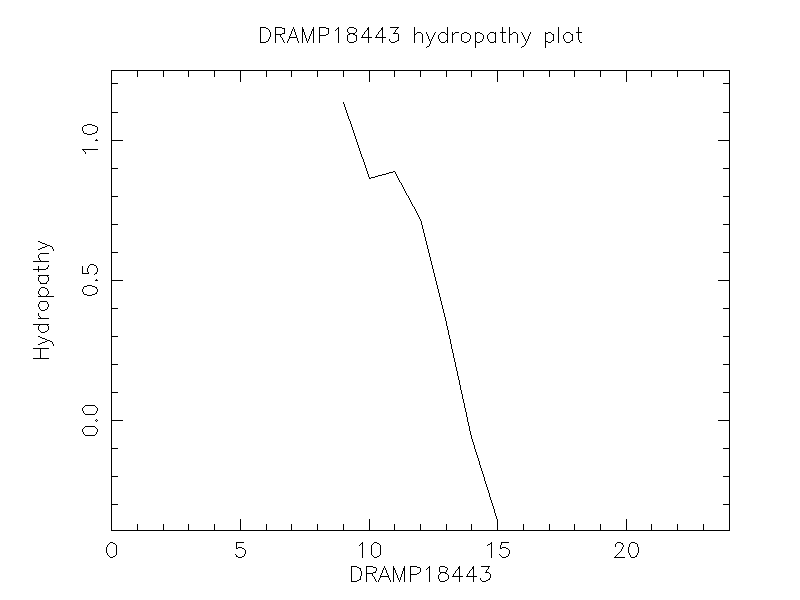
Hydrophobicity
- 0.313
Aliphatic Index
- 105.83
Half Life
-
- Mammalian:20 hour
- Yeast:30 min
- E.coli:>10 hour
Extinction Coefficient Cystines
- 5500
Absorbance 280nm
- 239.13
Polar Residues
- 7
DRAMP18443
Comments Information
Removal of the N-terminal four residues increases peptide cell selectivity.
Literature Information
- ·Literature 1
-
Title
- Characterisation of three alpha-helical antimicrobial peptides from the venom of Scorpio maurus palmatus
-
Pubmed ID
- 27019370
-
Reference
- Toxicon. 2016 Jul;117:30-6.
-
Author
- Harrison PL, Abdel-Rahman MA, Strong PN, Tawfik MM, Miller K.