General Information
-
DRAMP ID
- DRAMP18494
-
Peptide Name
- TP4
-
Source
- Oreochromis niloticus (Nile tilapia) (Tilapia nilotica)
-
Family
- Piscidin
-
Gene
- TP4
-
Sequence
- FIHHIIGGLFSAGKAIHRLIRRRRR
-
Sequence Length
- 25
-
UniProt Entry
- L0CKG3
-
Protein Existence
- Protein level
Activity Information
-
Biological Activity
- Antibacterial, Antifungal, anticancer, wound healing, Anti-Gram+, Anti-Gram-, Antimicrobial
-
Target Organism
-
- [Ref.29040295] Gram-positive bacteria: Staphylococcus aureus (MIC=8 μg/ml; MBC=16 μg/ml), Methicillin-resistant Staphylococcus aureus (MIC=16 μg/ml; MBC=32 μg/ml);
- Gram-negative bacterium: Pseudomonas aeruginosa (MIC=64 μg/ml; MBC=128 μg/ml);
- Fungi: Candida albicans (MIC=128 μg/ml; MBC=128 μg/ml)
-
Hemolytic Activity
-
- [Ref.29040295] 2% hemolysis at 0.78 μg/ml, 5% hemolysis at 1.56 μg/ml, 45% hemolysis at 3.13 μg/ml, 85% hemolysis at 6.25 μg/ml, 98% hemolysis at 12.50 μg/ml, 100% hemolysis at 25.00 μg/ml against mouse red blood cell
-
Cytotoxicity
-
- Not included yet
-
Binding Target
- bacterial outer membrane target protein, OprI-binding
Structure Information
-
Linear/Cyclic
- Not included yet
-
N-terminal Modification
- Not included yet
-
C-terminal Modification
- Not included yet
-
Nonterminal Modifications and Unusual Amino Acids
- Not included yet
-
Stereochemistry
- Not included yet
-
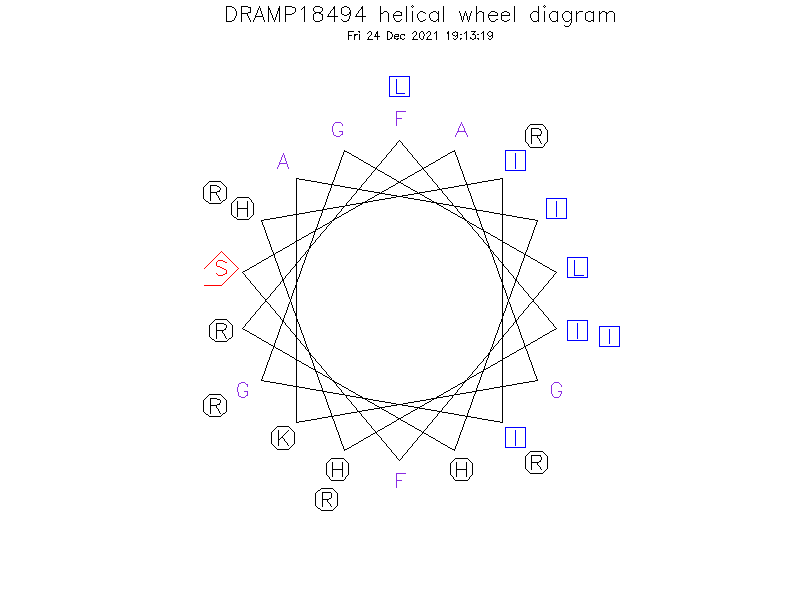
Structure
- Alpha helix
-
Structure Description
- 1.0x sarkosyl can drive TP4 into a conformational change from a non-structure to an alpha-helical structure, and that LPS can also drive TP4 to an alpha-helical structure but with high hydrophobicity and low solubility.
-
Helical Wheel Diagram
-
PDB ID
- 5H2S
-
Predicted Structure
- There is no predicted structure for DRAMP18494.
Physicochemical Information
-
Formula
- C135H226N50O27
Absent Amino Acids
- CDEMNPQTVWY
Common Amino Acids
- R
Mass
- 2981.6
PI
- 12.7
Basic Residues
- 10
Acidic Residues
- 0
Hydrophobic Residues
- 11
Net Charge
- +10
-
Boman Index
- -6561
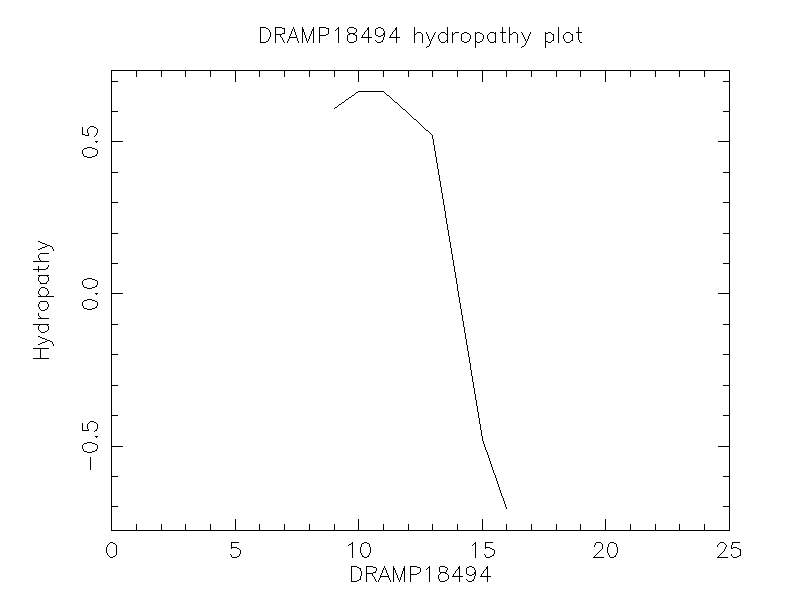
Hydrophobicity
- -0.128
Aliphatic Index
- 117.2
Half Life
-
- Mammalian:1.1 hour
- Yeast:3 min
- E.coli:2 min
Extinction Coefficient Cystines
- 0
Absorbance 280nm
- 0
Polar Residues
- 4
DRAMP18494
Comments Information
Function
- Antibacterial activity against the Gram-positive and Gram-negative bacteria. Antifungal activity. Has hemolytic activity.
Literature Information
- ·Literature 1
-
Title
- Hydrophobic residues are critical for the helix-forming, hemolytic and bactericidal activities of amphipathic antimicrobial peptide TP4.
-
Pubmed ID
- 29040295
-
Reference
- PLoS One. 2017 Oct 17;12(10):e0186442.
-
Author
- Chang TW, Wei SY, Wang SH, Wei HM, Wang YJ, Wang CF, Chen C, Liao YD.