General Information
-
DRAMP ID
- DRAMP21256
-
Peptide Name
- WL2 (Derived from CP-1)
-
Source
- Synthetic construct
-
Family
- Not found
-
Gene
- Not found
-
Sequence
- WLSKTAKKLWLSKTAKKL
-
Sequence Length
- 18
-
UniProt Entry
- No entry found
-
Protein Existence
- Synthetic form
Activity Information
-
Biological Activity
- Antimicrobial, Antibacterial, Anti-Gram+, Anti-Gram-
-
Target Organism
-
- [Ref.29266746] Gram-positive bacteria : Staphylococcus aureus ATCC 29213(MIC=128 μM), Staphylococcus aureus ATCC 43300(MIC=128 μM), Staphylococcus epidermidis ATCC 12228(MIC=128 μM), Bacillus subtilis CMCC 63501(MIC=64 μM);
- Gram-negative bacteria : Escherichia coli ATCC25922(MIC=8 μM), Escherichia coli UB1005(MIC=8 μM), Pseudomonas aeruginosa ATCC 27853(MIC=16 μM), Salmonella typhimurium ATCC 7731(MIC=32 μM), Salmonella pullorum C79-13(MIC=16 μM)
-
Hemolytic Activity
-
- [Ref.29266746] MHC5>128 μM against human red blood cells
-
Cytotoxicity
-
- [Ref.29266746] The cell viability of IPEC-J2 cells induced by WL2 is 99.0%, 95.0%, 89%, 89.8%, 85.2%, 86.8% and 75.1% at peptide concentrations of 2, 4, 8, 16, 32, 64 and 128 μM. The cell viability of IPEC-J2 cells induced by the control Melittin is 77.
-
Binding Target
- Not found
Structure Information
-
Linear/Cyclic
- Linear
-
N-terminal Modification
- Free
-
C-terminal Modification
- Amidation
-
Nonterminal Modifications and Unusual Amino Acids
- None
-
Stereochemistry
- L
-
Structure
- Random coil, alpha helix
-
Structure Description
- Not found
-
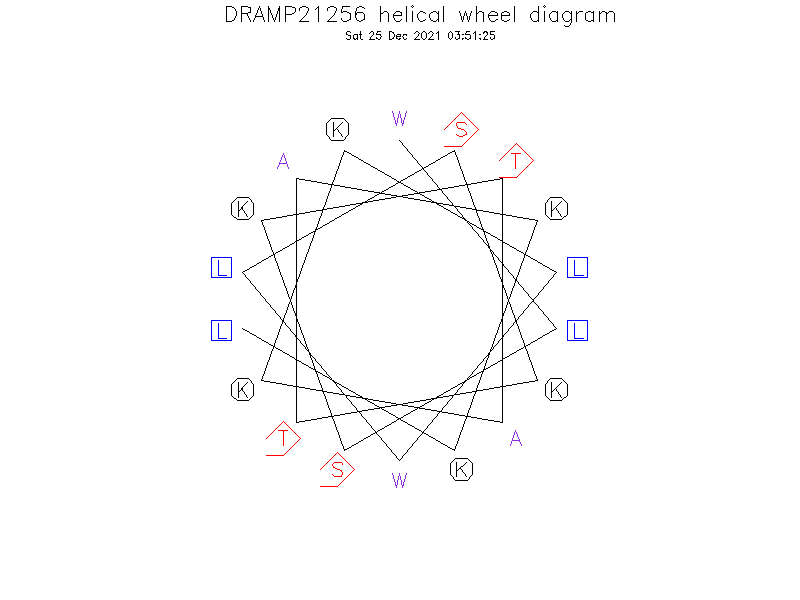
Helical Wheel Diagram
-
PDB ID
- None
-
Predicted Structure
- There is no predicted structure for DRAMP21256.
Physicochemical Information
-
Formula
- C102H172N26O23
Absent Amino Acids
- CDEFGHIMNPQRVY
Common Amino Acids
- K
Mass
- 2130.65
PI
- 10.7
Basic Residues
- 6
Acidic Residues
- 0
Hydrophobic Residues
- 8
Net Charge
- +6
-
Boman Index
- -1728
Hydrophobicity
- -0.522
Aliphatic Index
- 97.78
Half Life
-
- Mammalian:2.8 hour
- Yeast:3 min
- E.coli:2 min
Extinction Coefficient Cystines
- 11000
Absorbance 280nm
- 647.06
Polar Residues
- 4
DRAMP21256
Comments Information
Function
- Antibacterial activity against Gram-positive bacteria and Gram-negative bacteria
Literature Information
- ·Literature 1
-
Title
- Antibacterial activities and molecular mechanism of amino-terminal fragments from pig nematode antimicrobial peptide CP-1.
-
Pubmed ID
- 29266746
-
Reference
- Chem Biol Drug Des. 2018 May;91(5):1017-1029. doi: 10.1111/cbdd.13165.
-
Author
- Dong N, Wang Z, Chou S, Zhang L, Shan A, Jiang J