General Information
-
DRAMP ID
- DRAMP29028
-
Peptide Name
- Stripe
-
Source
- Synthetic construct
-
Family
- N/A
-
Gene
- N/A
-
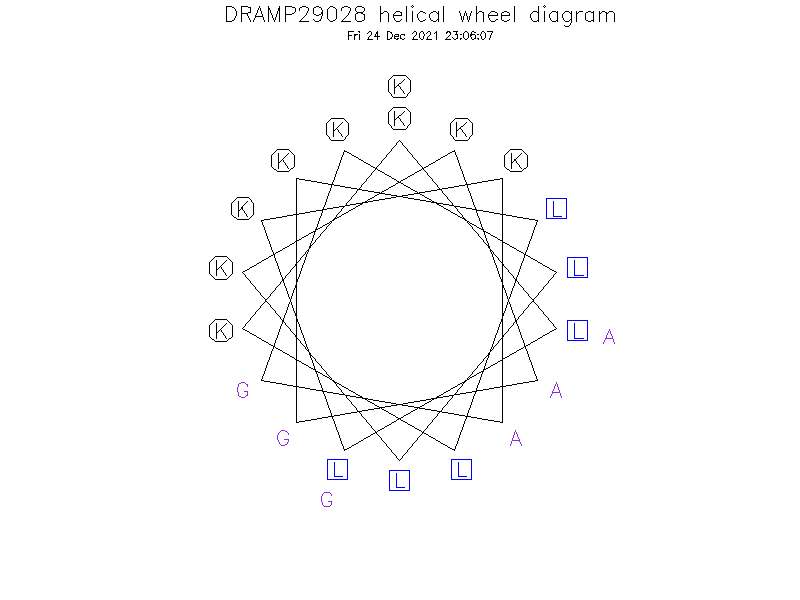
Sequence
- KLLKKAGKLLKKAGKLLKKAG
-
Sequence Length
- 21
-
UniProt Entry
- No entry found
-
Protein Existence
- Synthetic form
Activity Information
-
Biological Activity
- Antimicrobial, Antibacterial, Anti-Gram+, Anti-Gram-
-
Target Organism
-
- [Ref.33369262] Gram-positive bacteria: Staphylococcus aureus NBRC13276 (MIC = 12.5 μM) ;
- Gram-negative bacteria: Escherichia coli DH5α (MIC = 3.125 μM), Pseudomonas aeruginosa NBRC13275 (MIC = 3.125 μM), multidrug-resistant Pseudomonas aeruginosa ATCCBAA-2111 (MDRP) (MIC = 3.125 μM)
-
Hemolytic Activity
-
- [Ref.33369262] Stripe showed no hemolytic activity up to a concentration of 100 μM.
-
Cytotoxicity
- No cytotoxicity information found in the reference(s) presented
-
Binding Target
Structure Information
-
Linear/Cyclic
- Linear
-
N-terminal Modification
- Free
-
C-terminal Modification
- Free
-
Nonterminal Modifications and Unusual Amino Acids
- None
-
Stereochemistry
- L
-
Structure
- α-Helix content = 42% in 20 mM phosphate buffered saline (PBS) solution (pH 7.4), with 1% sodium dodecyl sulfate
-
Structure Description
- No detailed structure description found
-
Helical Wheel Diagram
-
PDB ID
- None
-
Predicted Structure
- There is no predicted structure for DRAMP29028.
Physicochemical Information
-
Formula
- C105H200N30O22
Absent Amino Acids
- CDEFHIMNPQRSTVWY
Common Amino Acids
- K
Mass
- 2234.93
PI
- 10.9
Basic Residues
- 9
Acidic Residues
- 0
Hydrophobic Residues
- 9
Net Charge
- +9
-
Boman Index
- -1218
Hydrophobicity
- -0.386
Aliphatic Index
- 125.71
Half Life
-
- Mammalian:1.3 hour
- Yeast:3 min
- E.coli:2 min
Extinction Coefficient Cystines
- 0
Absorbance 280nm
- 0
Polar Residues
- 3
DRAMP29028
Comments Information
Function
- Antibacterial activity against Gram-positive and Gram-negative bacteria.
The 'Stripe' is a rationally-designed amphipathic helical AMP comprising natural amino acids
- the AMP has hydrophobic Ala/Leu on one side of the helix and cationic Lys residues on the other. It has great antimicrobial activity and weak hemolysis. However, Stripe was compeletely degraded within an hour after incubation with proteinase K at 37℃, which means a bad stability.
Literature Information
- ·Literature 1
-
Title
- Rational Design of Helix-Stabilized Antimicrobial Peptide Foldamers Containing α,α-Disubstituted Amino Acids or Side-Chain Stapling
-
Pubmed ID
- 33369262
-
Reference
- Chempluschem. 2020 Dec;85(12):2731-2736. doi: 10.1002/cplu.202000749.
-
Author
- Motoharu Hirano, Chihiro Saito, Chihiro Goto, Hidetomo Yokoo, Ryuji Kawano, Takashi Misawa, Yosuke Demizu