-
-
-
-
-
-
Source
- Synthetic construct
-
-
Biological Activity
- Antimicrobial, Antibacterial, Anti-Gram-
-
- Function: Antibacterial activity against Gram-negative bacteria. Antibacterial activity against Gram-positive bacteria is not noteable under 100 μg/mL.
-
Target Organism
-
- [Ref.28547390] Gram-positive bacteria: Bacillus subtilis (MIC > 100 μg/mL), Staphylococcus aureus (MIC > 100 μg/mL), Staphylococcus epidermidis (MIC > 100 μg/mL);
- Gram-negative bacteria: Escherichia coli (MIC = 37.5 μg/mL), Shigella dysenteriae (MIC > 100 μg/mL), Salmonella typhimurium (MIC > 100 μg/mL), Klebsiella pneumoniae (MIC = 50 μg/mL), Pseudomonas aeruginosa (MIC = 25 μg/mL)
-
Hemolytic Activity
-
- [Ref.28547390] It has <1% hemolysis against human red blood cells at 25 μM and <1% hemolysis at 50 μM.
-
Cytotoxicity
-
No cytotoxicity information found in the reference(s) presented
-
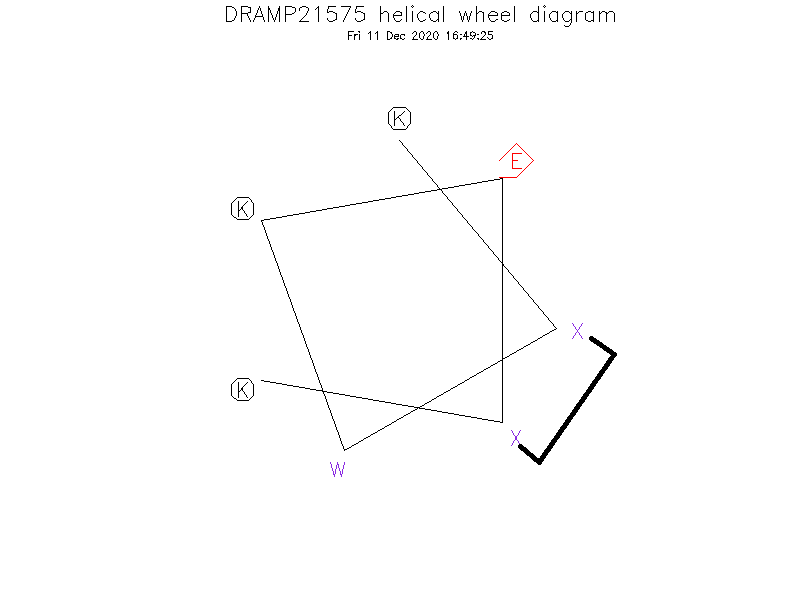
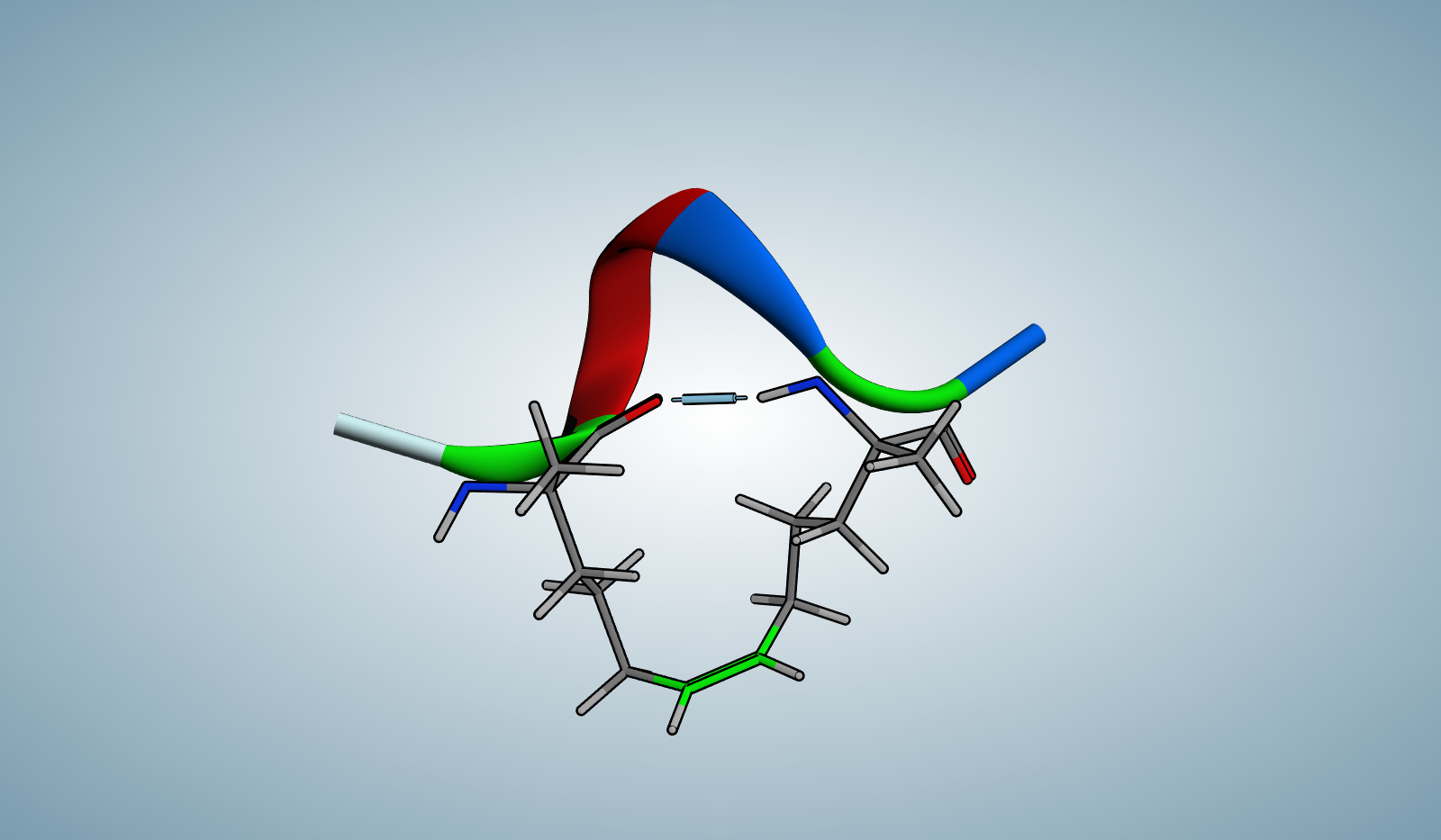
Linear/Cyclic
- Cyclic (Stapled)
-
N-terminal Modification
- Free
-
C-terminal Modification
- Amidation
-
Special Amino Acid and Stapling Position
- ①The Ⓧ (position: 2 and 6) in sequence indicates (S)-α-methyl, α-pentenylglycine. ②Ⓧ (2) and Ⓧ (6) are cross-linked by hydrocarbon stapling through an oct-4-enyl hydrocarbon staple.
-
-
Secondary Structure
- α-helix in a 25 mM potassium phosphate buffer solution at 20 ℃
-
Structure Description
- On the other hand, peptide PHE, TRP, and GLU, bearing phenylalanine, tryptophan, and glutamate, respectively, showed a slightly decreased helicity.
-
- Literature 1
-
Title
- Mono-substitution effects on antimicrobial activity of stapled heptapeptides
-
-
Reference
- Arch Pharm Res. 2017 Jun;40(6):713-719. doi: 10.1007/s12272-017-0922-1. Epub 2017 May 25.
-
Author
- Huy X Luong, Do-Hee Kim, Ngoan T Mai, Bong-Jin Lee, Young-Woo Kim